Galvanic corrosion can wreak havoc on piping systems and cooling equipment, ultimately triggering premature failure and unscheduled downtime. It’s not just a risk in heavy industry. Wherever two dissimilar materials touch in the presence of an electrolyte, galvanic corrosion can occur.
Everyday scenarios susceptible to galvanic corrosion include aluminum boats with steel fasteners, copper gutters mounted on a steel roof, and cars with aluminum bodies and steel frames. Industries such as HVAC, marine, and chemical processing have long designed systems for galvanic compatibility when specifying materials for pipes, hose, and fittings. More recently, the data center industry is becoming familiar with galvanic corrosion as mixed metal environments are needed to support emerging cooling technologies.
Understanding galvanic corrosion and how to control it is essential to maintaining long-term reliability in piping and cooling systems. This engineering bulletin explains:
- What galvanic corrosion is
- The conditions required for it to occur
- Where it appears in hose and piping systems
- Why it is increasingly relevant in data center cooling
- How operating conditions influence risk
- Practical design guidance to minimize exposure
What Is Galvanic Corrosion?
Galvanic corrosion is an electrochemical process in which one metal corrodes preferentially when two dissimilar metals are electrically connected in the presence of an electrolyte.
Galvanic corrosion, sometimes called dissimilar metal corrosion or bimetallic corrosion, occurs when current flows between two metals with different electrode potentials. The metals must be in direct contact. Water, salt solutions, humidity, and condensation are common electrolytes that allow this electrochemical reaction to occur.
When these conditions are present, a galvanic cell forms. The more active metal, the anode, corrodes at an accelerated rate as its atoms lose electrons and dissolve into the electrolyte as ions. The more noble metal, the cathode, is protected and corrodes more slowly or not at all.
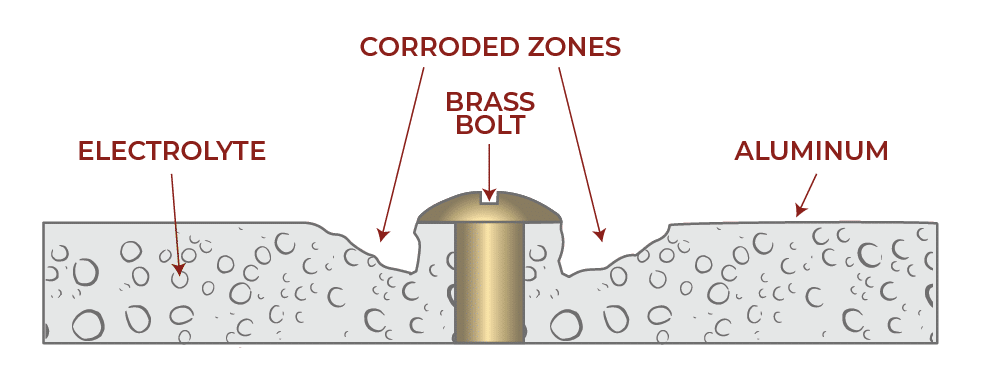
The illustration below shows galvanic corrosion in which aluminum sheet metal, acting as the anode, deteriorates around a brass bolt, the cathode.1
Understanding the Anodic Index
The relative potential of different metals, and the likelihood of galvanic interaction between them, are evaluated using an Anodic Index chart. The index assigns a numerical value in volts to metals based on their electrochemical behavior.
The larger the voltage difference between two metals, the greater the risk of galvanic corrosion. Design thresholds vary by environment and application, but the following guidelines are commonly used.
- ≤ 0.15 V difference: acceptable for harsh environments
- ≤ 0.25 V difference: acceptable for normal indoor environments
- > 0.25 V difference: increased risk of galvanic corrosion
The table below helps material and design engineers quickly identify metal pairings that may present galvanic compatibility concerns.2
Where Galvanic Corrosion Occurs in Piping Systems
Galvanic corrosion is a risk in piping systems constructed from multiple metals. For example, more than half of copper pipes and tubes produced globally are used in air-conditioning and refrigeration systems, meaning HVAC infrastructure relies heavily on copper.3 Other metals are also common. Aluminum is frequently used in heat exchangers, carbon steel appears in structural frames and supports, and stainless steel hoses are often selected when corrosion resistance is required. Valves and fittings are typically brass or bronze.
In mixed-metal environments like these, especially where moisture or other electrolytes are present, galvanic compatibility must be considered during both design and maintenance. Using the Anodic Index chart above, a materials engineer or facility owner evaluating common HVAC material pairings would come to the following conclusions.
While introducing stainless steel braided hose into a copper piping system may present some risk of galvanic corrosion, the operating environment ultimately determines whether the risk is significant. In systems operating at relatively low temperatures and circulating generally non-corrosive media with low chloride content, corrosion rates may be extremely low and well within industry limits for safe operation.
Galvanic Corrosion Risk in Liquid-Cooled Data Centers
As data centers move beyond traditional air cooling to direct liquid cooling, galvanic corrosion risk remains but the consequences of failure are far more severe. Even minor leaks or minute material degradation in liquid cooling loops can compromise sensitive IT equipment and disrupt critical processes.
Cold plates made from copper, flex hoses of stainless steel, and heat exchangers leveraging aluminum are a few common components in direct-to-chip liquid cooling systems. Coolant is typically a glycol-water mix or deionized (DI) water. The use of dissimilar metals and a continuously circulating electrolyte creates conditions where galvanic interactions can occur.
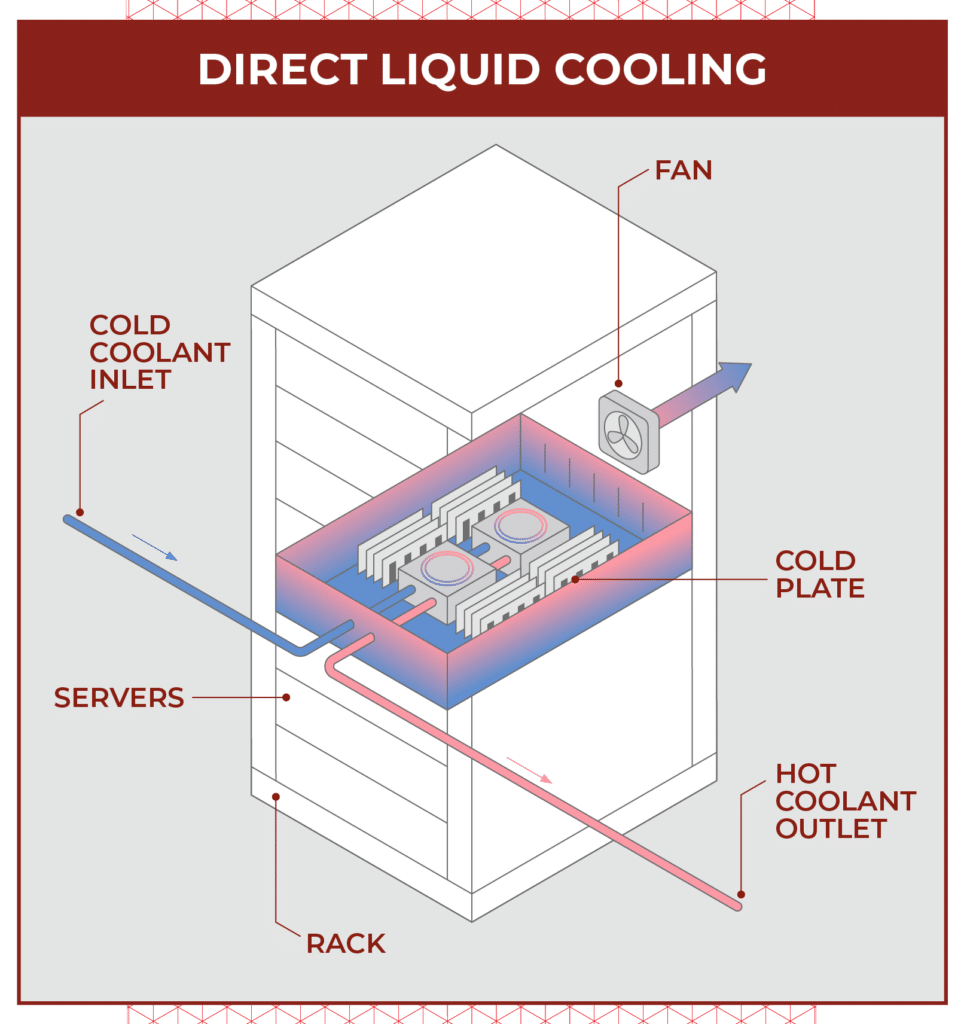
To mitigate this risk, system designers may consider alternative materials throughout the cooling loop. Hoses are one example. Though stainless steel is a popular choice for coolant conveyance, peroxide-cured EPDM is also used. While rubber hose itself is not a catalyst for galvanic corrosion, EPDM hose assemblies still feature stainless steel fittings. As a result, changing hose materials alone does not reduce the risk of galvanic corrosion in liquid cooled data centers where multiple dissimilar metal interfaces remain.
How Fluid Chemistry Influences Galvanic Corrosion
The electrolyte completes the galvanic circuit, and its conductivity, pH, and chemical composition play a significant role in determining how quickly corrosion progresses.
In liquid cooling systems, that electrolyte is typically a glycol-water mixture or deionized water. Higher glycol concentrations generally reduce conductivity, which helps limit galvanic reactions.
Additional protection is often provided by corrosion inhibitors such as silicates or phosphates, which passivate metal surfaces and form protective films. However, these additives must be carefully monitored and maintained. Over time, degraded glycol can oxidize and become acidic, lowering pH and increasing corrosion potential rather than suppressing it.
Because deionized water contains very few dissolved ions, its conductivity is low and galvanic activity is reduced. However, poorly maintained DI systems can become aggressive if contaminants accumulate and water chemistry drifts outside controlled limits. Dissolved chlorides and other impurities increase conductivity and disrupt protective oxide layers, accelerating corrosion.
Even in piping systems where water is not intentionally present in the process fluid, moisture can still act as the electrolyte. Condensation at metal interfaces is often sufficient to trigger galvanic interactions. For this reason, preventative maintenance is just as important as fluid chemistry management for long-term piping and cooling system reliability.
Galvanic Corrosion in Metal Hose Assemblies: Where It Occurs and How to Identify It
In metal hose assemblies, galvanic corrosion most commonly develops where dissimilar metals meet. Typical locations include weld joints where the hose, braid, ferrule, and end fitting connect, as well as points of contact between the hose assembly and adjacent equipment or piping.
The use of dissimilar metals within hose assemblies is not uncommon. For example, 316L hose is often paired with 304L braid. However, because these austenitic stainless steels sit close together on the Anodic Index chart, the risk of galvanic corrosion is minimal.
In other designs, particularly where fretting corrosion caused by vibration is a concern, a sacrificial layer of bronze braid may be introduced between the stainless steel hose and braid. In this case, the voltage difference between the metals is approximately 0.25 V. Under normal operating conditions, this difference may not present significant galvanic corrosion risk. However, in aggressive environments involving elevated temperatures, pressures, or corrosive media, the potential for galvanic corrosion increases.
In the field, galvanic corrosion typically appears as localized, rather than uniform, damage.
Common indicators include:
- Pitting concentrated near metal interfaces
- Accelerated corrosion on one metal
- Erosion at connection points
- Rapid material loss confined to a small area
In systems that include aluminum components, corrosion may appear as a white, powdery residue, a common byproduct of aluminum oxidation.
300 Series Stainless: Are Some More Vulnerable?
When evaluating galvanic corrosion risk among the 300 series stainless steels, the differences between grades such as 304L and 316L are relatively minor. Their Anodic Index values are very similar—approximately -0.58 V for 304L and -0.57 V for 316L in the active (non-passivated) state—meaning their galvanic behavior in a coupled system is nearly the same. While 316L offers improved resistance to certain forms of corrosion, particularly in chloride environments, it does not dramatically alter galvanic compatibility.
A more important distinction is whether the stainless steel surface is active or passive. Passivated stainless steels can exhibit Anodic Index values between approximately -0.08 V and -0.05 V, making them significantly more noble. The protective chromium-rich oxide layer substantially reduces corrosion susceptibility. For this reason, passivation can be used as a design control to meaningfully reduce galvanic corrosion risk in hose assemblies and piping systems.
Design Strategies to Reduce Galvanic Corrosion Risk
Beyond selecting compatible materials with minimal voltage differences, piping and cooling system designers must also account for environmental and operating conditions that influence corrosion rates. Effective galvanic corrosion control typically relies on a combination of material compatibility, electrical isolation, fluid chemistry management, and mechanical design considerations.
Operating Conditions
Temperature is one of the most significant factors. As a general principle, chemical reaction rates increase with temperature though actual corrosion rates vary by environment and alloy. Iso-corrosion charts demonstrate that corrosion acceleration is not linear and depends on specific chemistry and exposure conditions.
In high temperature applications featuring dissimilar metals and the presence of an electrolyte, designers must consider factors such as hose wall thickness, end fitting attachment method, and the use of coatings or additives in the flow media. Fluid monitoring should be an operational priority in these applications. Monitoring pH, maintaining inhibitor concentrations, and limiting contamination all help reduce corrosion risk.
Insulating Barriers and Protective Coatings
When dissimilar metals cannot be avoided, electrical isolation can reduce galvanic interaction. Insulating barriers such as non-conductive bushings, dielectric unions, or protective coatings interrupt electrical continuity and prevent the galvanic circuit from forming.
Protective coatings provide another layer of defense. Zinc plating, for example, offers sacrificial protection for carbon steel components, while stainless steel passivation enhances the stability of protective oxide layers.
Surface Area Relationships
Surface area relationships should also be considered during design. A small anodic component connected to a large cathodic structure will corrode more aggressively than if the surface areas were balanced. For example, a copper rivet in a steel sheet suffers little attack, whereas a steel rivet in a copper plate suffers very rapid attack.
Mechanical Stress
Finally, mechanical stress further contributes to risk. Vibration, thermal cycling, and system movements can disrupt protective oxide layers, introduce fresh electrolyte into contact areas, and expose bare metal at joints or interfaces. In dynamic systems, such as those using flexible metal hose assemblies, these effects can be more pronounced at weld connections and component interfaces.
Managing Galvanic Corrosion in Piping and Cooling Systems
Galvanic corrosion is predictable. Wherever dissimilar metals are electrically connected in the presence of an electrolyte, the potential for accelerated attack exists. As industries ranging from marine and chemical processing to semiconductor fabrication and hyperscale data centers adopt increasingly sophisticated cooling and fluid conveyance systems, the number of dissimilar metal interfaces continues to grow. With that growth comes elevated corrosion risk if compatibility is not carefully evaluated.
The good news is that galvanic corrosion can be managed effectively. Thoughtful material selection guided by Anodic Index values, intelligent system design, and disciplined fluid chemistry control all play essential roles. For metal hose assemblies, proper material selection, and fabrication practices are key to ensuring long-term corrosion resistance.
Understanding where galvanic corrosion begins is the first step toward preventing where it ends: leaks, contamination, unplanned downtime, and reduced system life.
Footnotes
[1] ATI Specialty Materials. High-Performance Alloys for Resistance to Aqueous Corrosion. ATI Specialty Materials, n.d.
[2] Scott Bredemann. “Galvanic Corrosion and Compatibility Chart.” MISUMI Mechanical Design Blog, updated April 8, 2025. https://us.misumi-ec.com/blog/galvanic-compatiblity-and-corrosion-table/
[3] Mordor Intelligence. Copper Pipes and Tubes Market – Growth, Trends, and Forecasts. Mordor Intelligence. https://www.mordorintelligence.com/industry-reports/copper-pipes-and-tubes-market
